Tuberculosis, a preventable and curable disease, ranks alongside HIV infection/AIDS as a leading cause of death worldwide. Indeed, new data from the World Health Organization reveal that the global tuberculosis burden is higher than previously estimated. The risk of developing tuberculosis is between 26 and 31 times greater in people living with HIV than among those without HIV infection. The existing vaccine is largely ineffective against pulmonary tuberculosis, the most common and contagious form of the disease. Drug-resistant strains of Mycobcaterium tuberculosis, the causative agent of the disease, continue to spread, far outpacing the development of new drugs. In the global context, tuberculosis in intimately linked to poverty, and control of tuberculosis is ultimately a question of justice and human rights. However, development of new diagnostics, therapeutics and vaccines is a global priority—numerous research efforts are currently underway, and point to possible successful outcomes.
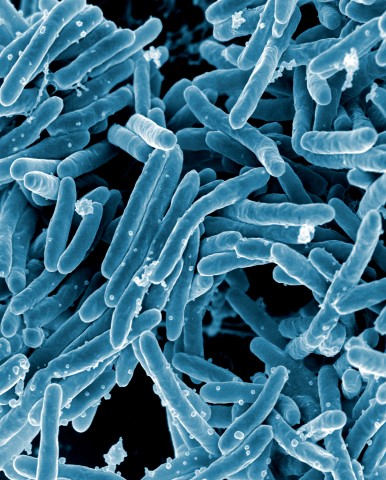
One of the new treatment options is based on the strengthening of the immune response, and the cellular process of autophagy has been proposed as a mechanism that the immune system uses to kill M. tuberculosis. Therefore, modulation of autophagy may represent a new option for the development of tuberculosis therapeutic and vaccine strategies, as indicated by research performed in mouse models. But is this approach safe? Results from a new study show that, in individuals with both tuberculosis and HIV infection, modulation of autophagy should be viewed with caution.
Robert Blomgran, senior author of the study, said in a press release: “When the world of research is looking for medicines, finding ways to strengthen immune defense is also now desirable, since medicines that attach the bacteria directly have been shown to lead to the development of resistance to antibiotics. Strengthening the immune defense system can be done through things like what is known as ‘stimulated autophagy’. But our research shows that the most common signal path for stimulating autophagy can be immediately dangerous.”
In the past few weeks, autophagy has been a newsworthy topic. Yoshinori Ohsumi has been awarded the 2016 Nobel Prize in Physiology or Medicine for his discoveries of its mechanisms. “Autophagy controls important physiological functions where cellular components need to be degraded and recycled. Autophagy can rapidly provide fuel for energy and building blocks for renewal of cellular components, and is therefore essential for the cellular response to starvation and other types of stress. After infection, autophagy can eliminate invading intracellular bacteria and viruses. Autophagy contributes to embryo development and cell differentiation. Cells also use autophagy to eliminate damaged proteins and organelles, a quality control mechanism that is critical for counteracting the negative consequences of aging. Disrupted autophagy has been linked to Parkinson’s disease, type 2 diabetes and other disorders that appear in the elderly. Mutations in autophagy genes can cause genetic disease. Disturbances in the autophagic machinery have also been linked to cancer. Intense research is now ongoing to develop drugs that can target autophagy in various diseases.”
The induction of autophagy has the potential to clear M. tuberculosis. Currently, pharmacological activation of autophagy is mostly achieved through what is known as “mTORC1 inhibition”. Blomgran and his collaborators were interested in its effects in presence of M. tuberculosis and HIV co-infection. The researchers found that in cells infected with a low dose of M. tuberculosis, stimulated autophagy through mTORC1 inhibition led to increased M. tuberculosis replication. In cells co-infected with M. tuberculosis and HIV, the increase in M. tuberculosis replication was even more pronounced.
Tuberculosis is present in either latent or active form. Individuals with latent tuberculosis infection do not feel sick and do not have any symptoms. They are infected with M. tuberculosis, but do not have tuberculosis disease, they are not infectious and cannot spread M. tuberculosis to others. Overall, without treatment, about 5 to 10% of infected individuals will develop tuberculosis disease at some time in their lives. For individuals whose immune systems are weak, especially those with HIV infection, the risk of developing tuberculosis disease is considerably higher than for patients with normal immune systems.
For their study, the researchers used a low dose of M. tuberculosis to mimic latent infection. Therefore, in their paper, they conclude that until proven otherwise, autophagy induction as a treatment option for tuberculosis should be handled with caution. Co-infection with HIV could increase the replication of M. tuberculosis and possibly activate tuberculosis in latently infected individuals.

The process of autophagy inhibition seems to have many important uses, including treatment of infection. The autophagy pathway is essential in eukaryotic cells to recycle cellular components for use in other compounds and structures. The mTORC protein, when activated, can set in motion a cascade of events leading to failure of autophagy. It seems that the mTORC protein also has implications in the natural aging process as the aging process can be hindered by the inhibition of autophagy through the mTORC pathway. Could it be possible that the mTORC pathway provides a research target for both immunological and age related studies? I really feel that these issues go hand in hand as natural immunity declines with increasing age. It would be great to “kill two birds with one stone”.
In regards to the utilization of autophagy there would be a need for studies to be done regarding different types intracellular pathogens to determine what methods, if any, they may possess that would allow them to survive or inhibit autophagy. The reason why the co-infection of HIV and M. tuberculosis is due to HIV inhibiting part of the autophagy pathway while M. tuberculosis is able to neutralize active nitrogen, thus leaving the intracellular M. tuberculosis under conditions for it to grow inside the cell. Cells use ubiquitination to select targets destined for autophagy and while there may pathogens like Salmonella enterica that are not able to neutralize the damage done by autophagy the processes utilized by HIV and M. tuberculosis prove that pathogens have evolve their own ways of getting around it. A wide use of methods that enhance autophagy could force an evolution of pathogens that develop ways to better inhibit the process or minimize the damages done by it, and lead us to have to develop new ways to fight those pathogens in the future similar to how we are having to do so now after over a century of antibiotics.
While there are several new drugs being developed for TB and treatments aimed at combating drug-resistant TB, it’s generally accepted that the days of antibiotics, and perhaps consequently drug treatments altogether, are coming to an end. The current alternative was introduced in 1921, nearly a century ago, and has its limitations. The scientific community has been charged with finding a modern alternative: one that will protect against all variety and strains of TB, one for people of all ages, and one for both the healthy and the immunocompromised, specifically with regard to HIV. As for the method covered in this blog, the application of any type of autophagy treatment sounds like it’s still in its infancy. In fact, any treatment that is established to coincidentally create more of the target pathogen sounds more like a gamble than an actual therapeutic at this point in time. For that reason, I can’t help but find myself drawn to what alternatives are currently under development. On that topic, there truly are a multitude of promising, upcoming vaccines in development. Vaccines based on virus-vectors, electroporated DNA vaccines, and use of heparin-binding haemaglutinin are all seeing development. Of all those types of vaccines, at least 14 are currently in clinical development.
Citation: BCG Vaccine | Current use & safety. TB Factsorg. http://www.tbfacts.org/bcg-tb-vaccine/. Accessed November 9, 2016.
Citation: Costa CD, Walker B, Bonavia A. Tuberculosis Vaccines – state of the art, and novel approaches to vaccine development. International Journal of Infectious Diseases. 2015;32:5-12. doi:10.1016/j.ijid.2014.11.026.
Shane,
I agree with your sentiment wholeheartedly. It appears the end of this golden age of antibiotics is quickly coming to an end. With that said, I respectively disagree with your dismissal of autophagy as a worthwhile avenue of study. As you stated vaccination is undeniably a vital tool in our collective repertoire. However, vaccines us our own immune system to confer resistance which in the evolutionary sense is static and can be seen as a limitation. I would argue why not attempt to improve out immune system with strategies such as autophagy manipulation?
Autophagy is a natural cellular process that can be effected to increase or decrease the level of orderly cell breakdown. Autophagy is necessary to replenish the proteins and cellular components needed to construct new cells. While we are still discovering the exact mechanisms that impact the process of autophagy, it is already very clear that it would be a good target for disease therapy. Beyond that, autophagy has been implicated in the natural aging process as activation of the autophagy pathway can lead to neurodegenerative disorders, cardiovascular disease, metabolic dysfunction and aging at an increased rate. Calorie restriction has shown to decrease the rate of autophagy, increasing life span. The differences in autophagy with aging were shown by researchers who looked at the genetic markers in young versus older brains and found a marked down regulation of the autophagy genes in the older brains. Autophagy is also a potent regulator of adaptive immunity and up regulation of autophagy could provide a means to therapeutically eliminate pathogens. (Augustine M.K. Choi, M.D., Stefan W. Ryter, Ph.D., and Beth Levine, M.D. Autophagy in Human Health and Disease.
February 14, 2013
N Engl J Med 2013; 368:651-662) Further research needs to be done to narrow down the specifics of autophagy-focused treatments but it seems that it has many potential uses and applications in fighting disease and improving immunity.
One thing that particularly interested me from this paper was finding that autophagy may be a mechanism responsible for some of the beneficial effects of caloric restriction. While I generally agree with Shane’s skepticism of autophagy, this paper does seem to indicate at least a ceiling on its utility, I can’t dismiss it’s use completely. Even this paper demonstrated that induced autophagy was effective on active mtb. What I suspect is that in the aftermath of antibiotic resistance the future of medicine will need a more complex, nuanced approach. To use a crude metaphor, if the age of firebombing antibiotics at pathogens is comparable to the use of fossil fuels for energy – a one size fits all solution, then the next age will be comparable to the multifaceted approach to energy we’ll probably have to use (solar, wind, nuclear, etc). Fighting disease in the future may require a range of innovations, with autophagy one part of the arsenal.
It is always interesting to monitor how our immune system play a role in stimulating the defense mechanism against pathogens and how the pathogens develops its tolerance level against immunity and develop new pathway to infect the host cells or organism. Sometimes it will be through mutation , sometimes through antibiotic resistance. Autophagy is the important defense mechanism of immune system to kill cancerous cells and virus or pathogen infected cells through a natural way . hence we all anticipated that enhancing autophagy can stimulated the immune system to fight against the infection . This study is interesting that the autophagy can actually help the TB to develop and replicate easily along with HIV infection TB cells can replicate better . This is a crucial finding in immunotherapy may be the mechanism and pathways of autophagy has to be studied closely and and its relation with the pathogen infection.There is a recent paper tells about the ayutophagyctivated by Bluetongue Virus Infection Plays a Positive Role in Its Replication. this is also a similar way which provoke the immune system and use them for enhancing its own replication in the cell.
Reference:
Autophagy Activated by Bluetongue Virus Infection Plays a Positive Role in Its Replication
Shuang Lv, Qingyuan Xu, Encheng Sun, Tao Yang, Junping Li, Yufei Feng, Qin Zhang, Haixiu Wang, Jikai Zhang, and Donglai Wu*
Arya,
It is fascinating how our immune system protects us from so many pathogens. Most of the time we don’t even know it’s working. All the different gasses and pollutants that we breathe in are demolished thanks to white blood cells. However, the occasional cold or flu viruses don’t let us take our immune system for granted. Every once in a while though, we encounter a virus or bacterium that requires medical intervention. TB is a disease that takes advantage our immune system and exploits its weaknesses. We need a functional vaccine or cure that can overcome the trickiness of this disease. As you mentioned, mutations and antibiotic resistant proteins allowed TB to reoccur throughout history. Autophagy is a natural physiological process that may have concurrent impacts on other immune and cellular responses. Although, it was discovered over 10 years ago, I don’t think the scientific community knows enough to manipulate its function. Therefore, I agree that autophagy should be studied carefully. I think more research should be done in respect to the role autophagy has in a specific disease. The mechanisms autophagy has at the time of exposure, infection and disease are important. Once these roles are discerned, maybe autophagy therapy could be used as a cure for multiple diseases and infections. However, patients still may be at risk of acquiring a genetic disease due to the disruption of autophagy via manual stimulation.
I agree with Shane in saying that even though models like this new study seem promising, there still is a lot unknown about the entire process and that these unknowns can be fatal for some of the most effected individuals. Like stated above there are plenty of alternatives that are in development but one of the problems they seem to have is creating viable models for their drug development that translate to the in vivo tests. Georgia State Chemistry department had a seminar speaker, Dr. Michael Niederweis, last spring that discussed their tests on Mtb. The first thing Dr. Niederweis’ group focused on was creating a drug efficiency testing model. In their new study published in August they’ve successfully created a Mtb/macrophage model that could be used in all these drug trials that are currently ongoing for tb. This model simulates the relationship between tb and macrophages and then allows researchers to inexpensively test their potential treatments in vitro, making this a very wide reaching study.
Another interesting thing that should be looked out for from Dr. Niederweis’ group was their current work he discussed briefly during his seminar where they are using their model to study the role of the outer membrane proteins on Mtb as potential treatment targets. They focused on previous findings that suggest tb uses iron to grow and spread and they studied the use of host’s heme by Mtb membrane proteins and how controlling it may lead to prevention and treatment. This is on going work that hopefully will have a great impact in the future. There is a plethora of studies being done, which is what we need to get rid of such a far reaching sickness.
Although Tuberculosis (TB) infections are a global priority, I disagree with the new autophagy modulation approach. According to the study mentioned in the original post, stimulated autophagy increased replication of TB in cells infected with HIV (1). The study also revealed that non-HIV cells of healthy patients had an increase in TB replication too (1). The researchers went on to hypothesize that autophagy stimulation decreased the acidification of the phagosome containing subcellular compartments and therefore supported mycobacterial growth (1). Based on the results of this study, I believe that autophagy, a natural immune response, should not be manipulated by pharmacological drugs to cure TB.
The mTORC1 inhibitor used to stimulate the autophagy response compromised the efficacy of the autophagy process. I think pharmacological drugs have unforeseen side effects in human cells. Although the drug may be designed to target a specific response, it may lead to degradation of normal cell processes. Inhibiting or activating cellular proteins could disrupt the initiation and formation of the autophagosome (2). This is because regulation of autophagy is controlled by a cascade of proteins and protein complexes (2). Therefore I think autophagy stimulation via drugs is too risky. Autophagy is an essential mechanism in our cells, and disruption in the autophagy process has been linked to Parkinson’s disease and other genetic diseases (2).
Hypothetically, if autophagy stimulation drugs were an approved treatment method for TB, then this would undoubtedly neglect the natural autophagy stimulation. Therefore, even though the TB patient gets cured, the disruption of natural autophagy could lead to their offspring being predisposed to a genetic disease by way of mutations in the autophagy genes.
“Autophagy induction targeting mTORC1 enhances Mycobacterium tuberculosisreplication in HIV co-infected human macrophages”. Scientific Reports 6, Article number: 28171 (2016) doi: 10.1038/srep28171. 15 June 2016.
1. “Autophagy induction targeting mTORC1 enhances Mycobacterium tuberculosis replication in HIV co-infected human macrophages”. Anna-Maria Andersson, Blanka Andersson, Christoffer Lorell, Johanna Raffetseder, Marie Larsson & Robert Blomgran. Scientific Reports 6, Article number: 28171 (2016) doi: 10.1038/srep28171. 15 June 2016.
2. “The 2016 Nobel Prize in Physiology or Medicine – Press Release”. Nobelprize.org.Nobel Media AB 2014. Web. 11 Nov 2016.
Porsha, I agree with your post 100%. The body’s natural immune response should not be tampered with in such a manner. “The mammalian target of rapamycin complex 1 (mTORC1) plays a fundamentally important role in the integration of metabolic, energy, hormonal, and nutritional stimuli to promote cellular biosynthesis” (1). Manipulation of this protein complex should not be done in people who are immunosuppressed. I was curious as to why this idea would have even been proposed in the first place and found that they’ve already cleared pharmaceutical drugs that target mTORC1, but only as a means of cancer treatment (2). This correlates to my argument because individuals with cancer have a rogue set of cells that are overreacting, dividing rapidly and taking extra nutrients from the body amongst other cancer phenotypical characteristics. They have proposed to use this drug in individuals who are immunosuppressed, their immune system is under performing, and the scientist are trying to remove a protein complex that is important for protein synthesis and producing energy. Further more the paper cited in the blog suggests that this potential treatment option should be “exercised great cautioned” based on findings from the single cell type that they used in the study (Human Monocyte-derived Macrophages), which is one of the many types of cells mTORC1 is present in and one of many cell types in the human body. I patiently await in vivo results.
1)Control of TSC2-Rheb signaling axis by arginine regulates mTORC1 activity… https://elifesciences.org/content/5/e11058
2)The mTOR Signalling Pathway in Human Cancer… https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3291999/
3)Autophagy induction targeting mTORC1 enhances Mycobacterium tuberculosis replication in HIV co-infected human macrophages… http://www.nature.com/articles/srep28171
4) Where is mTOR and what is it doing there?… http://jcb.rupress.org/content/jcb/203/4/563.full.pdf
Porsha,
I understand your point of view but I feel that we should not conclude anything based on just one study. The study mentioned in the blog post (1) shows that autophagy induction targeting mTORC1 enhances Mycobacterium tuberculosis replication in HIV co-infected human macrophages. However, contradictory results appear in another study (2). In this study, the researchers show that autophagy induction by vitamin D inhibits both Mycobacterium tuberculosis and human immunodeficiency virus type 1 in macrophages. The two studies ultimately induced autophagy although using different approaches, I think it depends on the mechanism used to induce autophagy that determines the final outcome.
Low vitamin D levels in human immunodeficiency virus type-1 (HIV) infected persons are associated with more rapid disease progression and increased risk for Mycobacterium tuberculosis infection. In this study, the researchers report that physiological concentrations of 1α,25-dihydroxycholecalciferol (1,25D3), the active form of vitamin D, inhibits M. tuberculosis and HIV replication in co-infected macrophages through human cathelicidin microbial peptide-dependent autophagy that requires phagosomal maturation. These findings provide a biological explanation for the importance of vitamin D sufficiency in HIV and M. tuberculosis-infected persons, and provide new insights into novel approaches to prevent and treat HIV infection and related opportunistic infections.
Citations:
1. Anna-Maria Andersson, Blanka Andersson, Christoffer Lorell, Johanna Raffetseder, Marie Larsson &Robert Blomgran. Autophagy induction targeting mTORC1 enhances Mycobacterium tuberculosis replication in HIV co-infected human macrophages. Scientific Reports 6, Article number: 28171 (2016)
2. Campbell GR, Spector SA. Vitamin D Inhibits Human Immunodeficiency Virus Type 1 and Mycobacterium tuberculosis Infection in Macrophages through the Induction of Autophagy. PLoS Pathog. 2012, 8(5): e1002689.
As alluded to in this blog post, tuberculous is a serious infectious disease of the respiratory track characterized by nodule-like growths. Historically human’s only recourse in combatting tuberculous was our immune system and its many avenues of defense. In the first half of the 20th century, however, the tools available in this fight changed drastically. The finding, understanding, and creation of anti-microbial agents gave us the upper hand against our microscopic advisories. For the past 70 years we have steadily increased our reliance on these antimicrobials, but putting all our eggs in this one basket may not have been the wisest course of action. With the emergence of antibiotic-resistant organisms, we must look for novel tools for the fight. Ironically, promising research relative to the improvement of host immunity such as autophagy may lead to fruitful discoveries. This work is vitally important to members of the immunocompromised community such as HIV patients because as mentioned in the post disproportionately suffer from a co-infection. But also may become a viable alternative to antibiotics generally. With that said, like any new technology, potentially dangerous outcomes must be addressed. One such concern is autography role in the proliferation of tumor growth which is briefly discussed by Zou P. They noticed an upregulation of mitochondrial autophagy in breast cancer tissue. Needless to say, the role this system plays in cancer is not well understood. Furthermore, it is important for us to perform our due diligence in this matter before we attempt any therapeutic intervention.
Citation: Zou P, Liu L, Zheng LD, Payne KK, Manjili MH, Idowu MO, Zhang J, Schmelz EM, Cheng Z. Coordinated Upregulation of Mitochondrial Biogenesis and Autophagy in Breast Cancer Cells: The Role of Dynamin Related Protein-1 and Implication for Breast Cancer Treatment. Oxid Med Cell Longev. 2016;2016:4085727. Epub 2016 Sep 26.
Carlos, this is a really interesting article. The fact that they found an upregualtion of autophagy can be linked directly to these cancer cells is a concerning point. But with this knowledge hopefully we can find a way to prevent these side effects when using it as treatment, especially since autophagy is gaining a lot of momentum as a treatment option for more than just tuberculosis. I found a recent review article looking into autophagy studies done in the past 10 years. It is in the context of traditional Chinese medicine so the methods of treatment are somewhat different than what western medicine has been looking into but it has a great comprehensive list of autophagy’s role in certain diseases and what upregulation and downregulation of autophagy can do for medicine. Over the years there has been a lot of promising results in the field even with all the unanswered questions. Because of the success more researchers are focusing on how to use it instead of studying what possible side effects it may have. It is important to consider everything and studies like the one you found may be vital for controlling the possible roadblocks with this treatment method, especially for patients who already have HIV and now tb, they can’t risk getting cancer too.
Citation: Wang, S.-F., Wu, M.-Y., Cai, C.-Z., Li, M., Lu, J.-H., Autophagy modulators from traditional Chinese medicine: Mechanisms and therapeutic potentials for cancer and neurodegenerative diseases. Journal of Ethnopharmacology.
Tuberculosis is caused by germ called Myobacterium Tuberculosis and can be spread through the air. While usually affecting the lungs, TB cab affect other parts of the body such as the CNS or the kidneys. Not everyone who becomes infected with the TB bacteria will become sick. The article discussed latent vs. active Tuberculosis. With latent Tuberculosis the individual does not have symptoms or feel sick. This person will have a positive skin test but cannot spread the infection to others. People with weak immune systems, especially those infected with HIV, risk developing the active version of TB. If TB overcomes the immune system the bacteria will begin to multiply and the person will become infectious to others and this condition can lead to death. Autophagy is believed to act in defense against intracellular pathogens. In humans genetic links between autopahgy, inflammatory bowel disease, and susceptibility to TB are all roles of autophagy.
Citation
“TB Elimination The Difference Between Latent TB Infection and TB Disease,” http://www.cdc.gov/tb. National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention: Division of Tuberculosis Elimination. November 2011. Accessed Nov. 2016
This article brought out the effects of autophagy on the immune system, especially its contributions towards HIV infected individuals. It made me ponder more about the catabolic process of autophagy. Autophagy is a catabolic process involving the degradation of cytoplasmic components, including whole organelles, via the lysosomal pathway. There are three different autophagic routes known; macroautophagy, microautophagy, and chaperone-mediated autophagy. In macroautophagy part of the cytoplasm to be degraded is wrapped inside a specialized organelle, then fuses with lysosomal vesicles delivering the engulfed cytoplasm for degradation. In microautophagy, the lysosomal membrane sequesters part of the cytoplasm. In chaperone-mediated autophagy proteins that have a specific sequence signal are transported from the cytoplasm through the lysosomal membrane, to the lysosomal lumen. Under stress conditions resulting cell death, autophagy is initially induced as an early prosurvival response before the death of a cell. Cell death by autophagy is cell death independent of apoptosis.
Citations
Denton D, Xu T, Kumar S. Autophagy as a pro-death pathway. Immunology And Cell Biology [serial online]. January 2015;93(1):35-42. Available from: MEDLINE with Full Text, Ipswich, MA. Accessed November 12, 2016.
Eskelinen, Eeva-Liisa, Saftig, Paul. Department of Biological and Environmental Sciences, Division of Biochemistry, University of Helsinki: Autophagy: A lysosomal degradation pathway with a central role in health and disease. Received 18 April 2008, Revised 9 July 2008, Accepted 10 July 2008, Available online 28 July 2008
Tuberculosis (TB) is one of the top 10 causes of death worldwide and it is a leading killer of HIV-positive people. People living with HIV are 20 to 30 times more likely to develop active TB disease than people without HIV. HIV and TB form a lethal combination, each speeding the other’s progress.
Due to indiscriminate use of anti-tuberculosis drugs, incorrect prescription by health care providers, and incomplete follow up of TB treatment regimen, drug resistant mycobacterium strains are emerging. Multi drug-resistant tuberculosis (MDR-TB) is a form of TB caused by bacteria that do not respond to isoniazid and rifampicin, the 2 most powerful, first-line anti-TB drugs. MDR-TB is treatable and curable by using second-line drugs. However, second-line treatment options are limited and require extensive chemotherapy (up to 2 years of treatment) with medicines that are expensive and toxic. Extensively drug-resistant TB (XDR-TB) is a more serious form of MDR-TB caused by bacteria that do not respond to the most effective second-line anti-TB drugs, often leaving patients without any further treatment options. So, I feel new research in the field of TB therapeutics is imperative. Given that autophagy plays a very important role in TB, research in this area seems to be a very promising option.
Besides its usual role in degradation of cellular proteins, autophagy is a cell-autonomous defense against intracellular pathogens including bacteria such as M. tuberculosis, Shigella, Salmonella, Listeria, and Streptococci or viruses. Autophagy kills intracellular mycobacteria by delivering to M. tuberculosis conventional anti-M. tuberculosis antimicrobial peptides such as cathelicidin through fusion with lysosomes where cathelicidin is stored, and by generating and delivering to M. tuberculosis a mixture of neo-antimicrobial peptides known as cryptides produced through autophagic proteolysis of innocuous cytosolic proteins such as ubiquitin and ribosomal proteins. Studies show that, several compounds that can induce autophagy without acting on the bacteria first have been shown to inhibit intracellular M. tuberculosis growth or survival. Thus, pharmaceuticals inducing autophagy could be potentially useful as new anti-M. tuberculosis chemotherapeutics.
Citations:
1. http://www.who.int/mediacentre/factsheets/fs104/en/
2. Bradfute SB, Castillo EF, Arko-Mensah J, et al. Autophagy as an immune effector against tuberculosis. Current opinion in microbiology. 2013; 16(3):355-365. doi:10.1016/j.mib.2013.05.003.
WHO states that at least one-third (1) of people living with HIV worldwide in 2015 were infected with TB bacteria. People living with HIV are 20 to 30 times more likely to develop active TB disease than people without HIV. In 2015 about 0.4 million people died of HIV-associated TB. About 35% of deaths among HIV-positive people were due to TB in 2015. In 2015 there were an estimated 1.2 million new cases of TB amongst people who were HIV-positive. Anti-TB medicines have been used for decades and strains that are resistant to 1 or more of the medicines have been documented in every country surveyed. Drug resistance emerges when anti-TB medicines are used inappropriately, through incorrect prescription by health care providers, poor quality drugs, and patients stopping treatment prematurely. Multidrug-resistant tuberculosis (MDR-TB) is a form of TB caused by bacteria that do not respond to isoniazid and rifampicin, the 2 most powerful, first-line anti-TB drugs. MDR-TB is treatable and curable by using second-line drugs. However, second-line treatment options are limited and require extensive chemotherapy (up to 2 years of treatment) with medicines that are expensive and toxic.
By boosting the immune system and using the cellular mechanism of autophagy can eliminate invading intracellular bacteria and viruses would decrease the gap of treatment in developed and developing countries. Though this form of treatment is still in its early stages and individuals with both tuberculosis and HIV infection should be viewed with caution when considering modulation of autophagy. This treatment would allow the host immune system to do the work for itself. Places such as India, Indonesia, China, Nigeria, Pakistan and South Africa that accounts for 60 percent of infection and poverty is high infected patients would be able to afford and keep up with this form of treatment. These countries are also known to have high rates of multi-drug resistance.
Citation:
(1) http://www.who.int/mediacentre/factsheets/fs104/en/
The rise of antibiotic resistance has led to a scramble to find alternative methods of combating diseases recently thought somewhat under control. Manipulation or amplification of processes already in place is a potential avenue of attack and autophagy specifically has shown some promise. This study however demonstrates that there are considerable risks, with induced autophagy resulting in replication of latent TB (1). This risk is possibly common with intracellular pathogens, as autophagy was found to spur replication of Salmonella as well (2). There may be limitations to autophagy’s utility, with its use dependent on context i.e. as a last resort when pathogens such as TB are in an active state. As evolutionary mechanisms “created” this problem I have wondered if evolutionary mechanisms could be exploited in attacking disease. Though there are notable exceptions, a high fitness cost due to drug resistance has been found in many pathogens including M. tuberculosis (3). In particular it’s been demonstrated that bacterial pathogens that have acquired drug resistance often have lower growth rates and are less transmissible than drug susceptible variants (4). Exploiting these fitness costs could be a potential means of eradicating or at least managing these diseases.
Citations
(1) Andersson, AM, Andersson, B, Lorrell, C, et al. Autophagy induction targeting mTORC1 enhances Mycobaterium tuberculosis replication in HIV co-infected macrophages. Sci Rep 6, Article number 28171 (2016). doi:10.1038/srep28171
(2) Citation Yu HB, Croxen MA, Marchiando AM, et al. Autophagy facilitates Salmonella replication in HeLa cells. mBio 5(2):e00865-14. (2014) doi:10.1128/mBio.00865-14.
(3) Melnyk, A., Wong, A. & Kassen, R. The fitness costs of antibiotic resistance mutations. Evol. Appl 10.1111/eva.12196 (2014)
(4) Schulz zur Wiesch, P, Engelstadter, J, Bonhoeffer, S. Compensation of fitness costs and reversibility of antibiotic resistance mutations. Antimicrob agents chemother 54(5): 2085-2095. doi:10.1128/AAC.01460-09.
The topic of autophagy and its importance to Immunosurveillance is a topic that could ultimately find a resolution to the ongoing issue of TB infection and even go as far as to address other topics such as caner immunology. Auotophagy is commonly triggered by damage-associated molecular patterns (DAMPs), which can originate from a multitude of sources. DAMPs, also known as danger-associated molecular patterns and danger signals, are host biomolecules that can initiate and perpetuate a noninfectious inflammatory response. In contrast, pathogen-associated molecular patterns (PAMPs) initiate and perpetuate the infectious pathogen-induced inflammatory response (2). Immunosurveillance is a term used to describe the processes by which cells of the immune system look for and recognize foreign pathogens, such as bacteria and viruses, or pre-cancerous and cancerous cells in the body. Immunosurveillance and immunoediting is based on protection against development of spontaneous and chemically induced tumors in animal systems and identification of targets for immune recognition of human cancer (1). This topic is related to autophagy in that it precedes immunosurveillance in the case that bacteria, viruses, and cancerous cells are found. The necessity of autophagy and its relation to the cure of TB has also been found to be pivotal in the regression of tumors by increasing the prevalence of specific tumor associated antigens (TAAs) which leads to increased autophagy of the TAAs and increased immunological attack of tumor cells.
(1) Dunn, G. P., Old, L. J., & Schreiber, R. D. (2004). The immunobiology of cancer immunosurveillance and immunoediting. Immunity, 21
(2), 137-148.
Bianchi, M. E. (2007). DAMPs, PAMPs and alarmins: all we need to know about danger. Journal of leukocyte biology, 81(1), 1-5.