Macrophages—the big eaters of the immune system—reside in most tissues. They’re plastic, and change their function depending on the environmental cues they receive. Therefore, they play many different roles. Results from a new study highlight one of these new roles, until now unknown—macrophages grab the broken ends of blood vessels and stick them back together.
The study (Macrophages Mediate the Repair of Brain Vascular Rupture through Direct Physical Adhesion and Mechanical Traction) was carried out in zebrafish, which served as model of human brain microbleeds. The occurrence of microbleeds is a pervasive problem in older individuals. “The brains of older people are sometimes peppered with dark splotches where blood vessels have burst and created tiny dead zones of tissue.” In the general population, a high microbleed count is associated with an increased risk for cognitive deterioration and dementia, and appears to mark the presence of neurodegenerative brain damage.
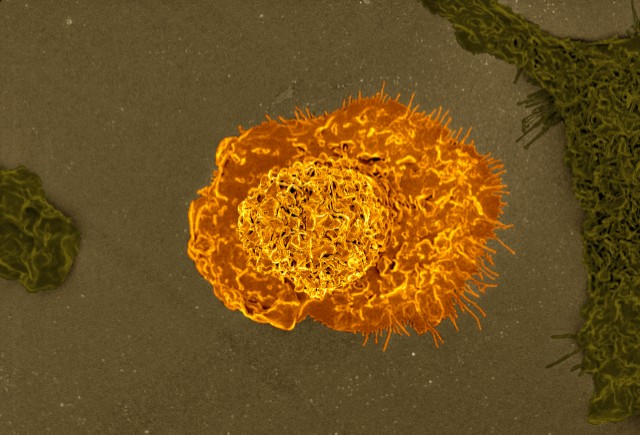
For the study, researchers used lasers to create a clean split with two broken ends in the small blood vessels of the zebrafish brain. Then, through a specialized microscope, they observed what happened after the blood vessels were ruptured. The researchers noticed an unexpected repair process that started about a half hour after they induced the vessel damage. A macrophage showed up, and extended two “arms” from its body toward the ends of a broken blood vessel, using a variety of adhesion molecules to attach itself to the vessel. Then, it pulled the two broken ends together to mediate their repair. The macrophage left the scene after the blood vessel was repaired. The whole process took about three hours.
In addition, the researchers noted that usually only one macrophage arrived at the laser-wound site to mend the broken ends. Rarely, two macrophages arrived at the injury on their own—in these instances, each macrophage grabbed one of the broken ends of the blood vessel. However, the macrophages then disengaged from the vessel without fixing the damage.
Chi Liu, lead author of the study, said in a press release: “At the beginning, we weren’t sure this was a repairing behavior. After we confirmed that the macrophage mediates this repair through direct physical adhesion and generation of mechanical traction forces, we were excited. This is a previously unexpected role of macrophages.”
The researchers observed a similar repair process even for a ruptured blood vessel in the zebrafish fin. After the blood vessel was ruptured using the laser, a macrophage arrived at the injury site and extended its protrusions to pull the broken ends of the blood vessel back together.
Luo added: “Several aspects of vascular development and remodeling, associated with macrophages, are conserved in human and zebrafish. Microglia [a subset of macrophage cells] are required for the repair of blood-brain barrier injury in mice, and macrophages can be found surrounding most capillary microbleeds in humans. We believe that the macrophage repair system in our study is very much likely replicated in humans and mice.”
Watch this video to see how, after cerebrovascular rupture, a macrophage (green) migrates to the lesion, extends protrusions (arrowhead), adheres to and pulls the ends of the blood vessel (orange) together, and finally leaves the lesion when the repair is accomplished. Duration of imaging is 285 minutes.

I find it very interesting that macrophages have another function, beside the uptake of infected cells, which is to repair blood vessels and other tissues. It has already been reviewed that the increase of macrophages at the site of repair will leave the broken blood vessel as is, and the repair will not be completed. Because the study noticed that the repair of the cut blood vessels can only be repaired by one macrophage, which takes about 3 hours to complete, I wondered if it was possible, with the use of enzymes or specified cytokines, that the speed of the repair of a single broken blood vessel would increase. Is it possible that the researchers are currently looking at ways in which the repair of the broken blood vessels can increase in speed? Also, if there are more than one broken blood vessel in a localized area, is it a first come first serve opportunity for the macrophages? Or will there be many non-repaired vessels due to an overabundance of macrophages in the area?
I approached the topic in the same manner. I found that there are some studies currently using macrophage therapy in regenerative medicine, which to me is fascinating. I pondered the same question and wondered if there were a way to find exactly what cytokine recruits these macrophages, and could increasing it’s concentration be an effective treatment? Someone else mentioned that simply increasing the concentration of macrophages in the system may not result in any significant change since as you stated -only one macrophage can do the job. Perhaps in those with dimentia, their macrophages are just dysfunctional and a treatment with normally functioning macrophages could do the trick!
Interesting post. When I think about blood vessel repair my immediate thought is the classic image of an aggregation of platelets wrapped in a fibrin mesh. So I was surprised to learn that macrophages, which are immune cells, could also function directly in blood vessel repair. I was also impressed with the speed and efficiency of their repair, relative to other mechanisms. My impression after reading the summary is that macrophages act as “placeholders” or temporary fixes in compromised blood vessels, until more cells (e.g. platelets, coagulation proteins, effector cells) can be recruited. My thinking is that macrophages act primarily as phagocytic cells, and as the first effector cells present in tissue injury, they can also secrete cytokines which enhance inflammatory response and thus blood vessel repair. In the case of small blood vessels, macrophages can singly reattach them and accelerate healing. Like the other commentators, I am not sure why only one macrophage can repair a blood vessel at a time, other than the ridiculous notion that just popped into my head after watching the video, namely that the second macrophage would adhere to the first and somehow get stuck during the repair. Anyways, this study really highlights the versatility of macrophages, and it (specifically the direction the research is going towards) offers a lot of hope in medicine I think. Tissues that we thought were not capable of healing due to ischemia (e.g. the brain, nerves) may actually be successfully repaired if we know more about the mechanisms behind blood vessel repair, of which macrophage-mediated blood vessel repair is only one. As Cattin et al. (2015) reports, macrophages can sense hypoxia in peripheral nerves; in response, they produce vascular endothelial growth factor A (VEGF-A), and they use the newly-created blood vessels to direct nerve regeneration. Fascinating!
Cattin A-L, Burden JJ, Van Emmenis L, et al. Macrophage-Induced Blood Vessels Guide Schwann Cell-Mediated Regeneration of Peripheral Nerves. Cell. 2015;162(5):1127-1139. doi:10.1016/j.cell.2015.07.021.
This was such a great read. This article is a prime example of how science is always evolving which helps young scientist, like myself, keep a full time job researching new discoveries. Prior knowledge of macrophages says that macrophages main job is to locate and eliminate foreign pathogens that may cause harm to the body. Scientist are discovering that macrophages many different functions, one being the ability secrete anti-inflammatory cytokines, which promote angiogenesis, the process through which new blood vessels form from pre-existing vessels , this is referred to as possessing an “alternative” phenotype. “The alternative M2 macrophage activation pathway typically deactivates macrophages after exposure to Th2-type cytokines. Such responses characterize immunoregulatory, immunosuppressive, and protumoral settings. M2 cells induced by exposure to IL-4 and IL-13 (M2a) and deactivating cytokines such as IL-10 and TGF-β (M2c) are thought to suppress immune responses and promote tissue remodeling ” (Richardo,2008) .This statement gives a visual of how the “alternative” M2 macrophage promotes remodeling and repair in the body.
Sharon D. Ricardo, Harry van Goor, Allison A. Eddy “Macrophage diversity in renal injury and repair” Published November 3, 2008
You definitely bring up a good point that science is always evolving from the more advanced technology and resources we have available to us. It’s up to society to utilize these collaborative efforts in working towards healthier lives. I also found it fascinating that researchers have been able to observe other behavioral characteristics of macrophages. In a more recent study, they concluded that both microglia and macrophage phenotypes are highly important for CNS restoration in reaction to severe injuries. The adult brain was thought to be inactive after injury but enough evidence has shown that there is still some degree of plasticity in this organ after an injury. It is known that activated microglia and macrophages with discrete phenotypes have the ability to stimulate or inhibit neural revival after a patient suffers from any neurological debilitating injury or disorder. More specifically, the M2 cells have the capability to aid with neurogenesis, axonal remodeling, angiogenesis, oligodendrogenesis, and remyelination. With this information at hand, it seems to be possible for target drug treatments using this mechanism. It will be interesting to see the developments in therapeutic deliveries for stroke and other similar injuries to the nervous system in the coming years.
Hu, X., Leak, R. K., Shi, Y., Suenaga, J., Gao, Y., Zheng, P., & Chen, J. (2014). Microglial and macrophage polarization—new prospects for brain repair. Nature Reviews Neurology Nat Rev Neurol, 11(1), 56-64. doi:10.1038/nrneurol.2014.207
I found this post to be extremely interesting and it actually sparked my interest to research and investigate other roles macrophages play in the human body. I came across an article from The Atlas of Science that talks about how electric fields drive the healing process of macrophages. It’s already widely known, for example, that the signals from cytokines control the functions of macrophages. What there is not a lot of information about, however, is how a macrophage responds to the “physical stimulus of an electric field.” The article shows that electric fields can influence the direction in which a macrophage migrates. They say the cathode is the wound’s center and the anode is the edge of the wound. Macrophages tend to move toward the positive pole (edge of wound) when there is an electric field where they will make chemical mediators to tell nearby and neighboring cells to speed up and help the healing process. I am wondering if anyone else has heard of this before and can provide more details about this process?
This information is really interesting, I wonder how and why the researchers decided to study this. Obviously they were able to see repairs of the brain microbleeds by macrophages in the zebra fish, but are unable to examine this process in humans yet. However, if the elderly are experiencing dementia or cognitive deterioration, maybe those individuals have macrophages that are unable to fuse the blood vessels back together, or not enough are able to be repaired. I was unable to find an answer to this, but I also don’t think macrophage therapy or increasing macrophage levels would be beneficial considering that multiple macrophages at one broken blood vessel do not fix the damage.
I decided to do some research on how to prevent brain microbleeds, and it appears that a lot more research needs to be done to better understand the mechanism behind them. Dr. Mark Fisher describes how “newer medications interfere with blood clotting (to protect against stroke) while at the same time protecting the blood vessel wall (to help prevent bleeding) and he suggests that MRI screening be used more strategically to identify patients with microbleeds, allowing their physicians to adjust treatments accordingly.”
It’s also possible that high blood pressure could be responsible for the microbleeds and because the aorta stiffens with age, more pressure is exerted on the brain strong enough to burst vessels. It’s possible that reducing the stiffness of the aorta with antioxidants or fatty acids may help prevent these microbleeds.
Who would have thought!? But should we really be surprised? After all, macrophages are always lurking around. I completely agree with the fact that high blood pressure may have a role in microbleeds, since the brain receives 15% of the cardiac output, as stated in the article linked above. Also, it seems like there is a connection between blood pressure and aortic stiffness, since those who have both do not do as well on cognitive assessments dealing with visual processing and memory. However, more studies need to be done. So as you stated, although macrophage therapy would not be a viable option, maybe the best way to deal with microbleeds is simply managing the health of oneself.
Discovering that macrophages have the ability to repair broken blood vessels is completely new and interesting information to me. The article mentioned that a “high microbleed count can lead to dementia.” I immediately thought about my aunt who has early stages of Alzheimer’s disease. This led me to question: If macrophages can repair blood vessels, are they performing this role in Alzheimer’s disease? If so, why does this disease continue to progress? In a published study by Vincent Marchesi, it highlighted that Alzheimer’s disease and CADASIL (damage of the arteries) are not only caused by amyloid deposits, but by damaged, small blood vessels of the white matter regions in the brain. The damage in these small blood vessels is caused by a mutation in the NOTCH3 gene. When NOTCH3 is mutated it damages the blood vessels in the white matter regions of the brain, thus leading to Alzheimer’s disease, dementia, etc. Now that we know the new function of macrophages, how does this repairing process occur in individuals with these diseases? In the article, “Cyclooxygenase-2-positive macrophages infiltrate the Alzheimer’s disease brain and damage the blood–brain barrier,” discovered that only CD68 macrophages enter the perivascular space. However, most macrophages do invade the amyloid-β plaques, but only surround the vessels that contain the amyloid deposits. From this we can conclude that maybe this is the reason why Alzheimer’s disease continues to progress and get worst in individuals who have it.
References:
Fiala, M., Liu, Q. N., Sayre, J., Pop, V., Brahmandam, V., Graves, M. C. and Vinters, H. V. (2002), Cyclooxygenase-2-positive macrophages infiltrate the Alzheimer’s disease brain and damage the blood–brain barrier. European Journal of Clinical Investigation, 32: 360–371. doi: 10.1046/j.1365-2362.2002.00994.x.
Marchesi, V.T. Cell. Mol. Life Sci. (2014) 71: 949. doi:10.1007/s00018-013-1542-7.
The activity of Macrophages, white blood cells that are responsible for engulfing pathogens in the body, aren’t well studied as their full potential is still unknown. These cells play an important role in wound healing including repairing blood vessels as mentioned in the article above. According to recent studies , macrophages reduce inflammation while promoting wound healing through various actions that may be of interest for relieving pain in the near future . According to this research article, macrophages exist in several “phenotypic states “which allow them to differentiate and accomplish multiple tasks at once. These cells, are part of the body’s first line of defense and play a critical role in both acquired and innate immunity. As science further develops, it is interesting to see how these white blood cells play several roles in our body. This article is extremely fascinating since it reveals new information about macrophages, as engulfing of pathogens through phagocytosis is the most well known function of these immune cells.
This was interesting information regarding the ability of macrophages to have a function of repairing broken blood vessels in the body. This can have numerous implications for various diseases that cause degeneration of blood vessels in various parts of the body. It is surprising that only one macrophage is capable of mending a blood vessel. I cannot help but wonder if this is efficient for the body in mending blood vessels and the reasoning behind the lack of more macrophages to do the same task. I also think about the role of macrophages in a tumor suppressive environment. Based on a study conducted by Lewis et al., perivascular tumor-associated macrophages (TAMs) are implicated in angiogenesis, cancer cell invasion and also cancer growth. A high number of TAMS is correlated with a lower survival rate in numerous types of cancers. Undoubtedly, great therapies will result in learning their function in repair for the vascular system, yet other therapies to target overstimulation of these functions should also be considered. The newfound function of macrophages definitely has great implications in therapies that will help those that suffer from hemorrhages in their blood vessels, yet the potential for macrophages to progress a disease should also be realized.
References:
The Multifaceted Role of Perivascular Macrophages in Tumors.
Cancer Cell. 2016 Jul 11 ;30(1):18-25. doi: 10.1016/j.ccell.2016.05.017.
For some reason, this article didn’t come to me as a surprise, impressive yes, because these brilliant researchers were able to figure this process out. Why not surprising? If you think about it truly, macrophages are there to help the body fight infections by recognizing PAMPs (pathogen-associated molecular patterns), releasing cytokines to call for help and by helping activate the adaptive immune response. Macrophages have to recognize something before they react; although they did not go through the same process as lymphocytes in secondary lymphoid organs, they still behave the same way in the sense that they do not attack cells or particles that belong to the body in which they reside. When endothelial cells of a blood vessel are intact and fully joined together, that blood vessel has nothing new that macrophages have never seen; however once there is a cut the cells change shape and structure resulting in sharp edges formation making them now a new thing. I believe PAMPs are not only pathogen related, they include anything that represent a threat to the body and a cut blood vessel is one. Cancer cells used to be spared by cytotoxic T cells until they became cancer, thus disturbing the natural state of our body causes reactions. No cell or even organ in our body can stand alone; it is a joint effort and collaboration by all that maintain a human body. Finally, this tells us how much there is to be found out. Despite the big progress science has made over time; the unknown is still outweighing the known. As the saying goes, knowledge is power and with the rate at which new discoveries are appearing almost daily, there is hope that one day scientist will get to find cures to diseases that are believed to be incurable today, diseases such as HIV, cancer etc.
I liked how this article described macrophages as the “big eaters” of the immune system. I did not know that macrophages serve other functions which fascinates me to learn more about the secret life that they have. Macrophages can repair blood vessels that are broken in the body is a fact that interested me. I am happy to see that they tested this fact with the zebrafish model, showing in an unexpected way that macrophages can repair ruptures through direct physical and mechanical traction.
I read this article about how macrophages play a role in the inflammation in Atherosclerosis which is a disease in which plaque is built up in the arteries. Interesting fact about this article is that macrophages can export cholesterol, secreting it through ATP-binding cassette transporters such as ABCA1. This an important step because accumulation of cellular cholesterol can help in repair the plaque in the arteries. The article also talks about M1 macrophages and M2 macrophages in greater detail which shows that science can be discovered in advanced ways. Soon in the future we can learn in depth what else can macrophages do in a different way.
Red blood vessels are found all through the body, but the repair of red blood vessels in the liver aids in disease recovery. Macrophages can be used for liver repair by repairing broken blood vessels. The depletion of certain macrophages can lead to delayed disease recovery. The liver is used for detoxifying and metabolizing drugs, so without proper functioning liver the disease will persist. If macrophages are able to repair blood vessels in the liver, then the liver can properly metabolize drugs and detoxify the blood. With the macrophage blood vessel repair mechanism, a disease like fibrosis can become repairable.
This article was so interesting in the fact that macrophages have not done this before in prior research. This article makes me think of how macrophages are the first cells to show up when you start to get an infection during the innate immunity and they start to induce an inflammatory response. In similarity, when the blood vessel ruptures they are the first ones to show up and help repair the two vessels together. I think it is also interesting how there is only one macrophages used in the blood vessel repairing process. The article states when there is two macrophages at a time, the damage usually does not get repaired. Would this be because the body can’t handle having more than one macrophages around for this particular incident?
I think the idea that macrophages may be one of the main ways to repair ruptured blood vessels means that macrophages are much more than purely engulfing cells. It seems as if macrophages since they are situated near capillary microbleeds in humans as if they are just prepared for something like this, but we have just never noticed that they could do this before. Further research of course needs to be done to gain more insight into the situation at hand, but it from the looks of it macrophages are a much more prominent cell than just the initial cell of the innate immune system.
I found this blog post fascinating since my grandfather passed away by a hemorrhagic stroke. The study demonstrated that macrophages repair the brain of microbleeds in zebra fish, which is promising and hopefully, leads to a human study. This post sparked my interest in finding other possible studies that possibly being conducted regarding stroke improvement as it relates to innate immunity. One, study that really captivated me was looking at natural killer cells (NK) in ischemic stroke patients which can used to identify as a biomarker for risk in developing infections. NK cells are the first part of the innate response against infection of cells. The study showed that NK cells was in abundance within the first few hours after the stroke occurred that led to infections. The study also showed that a brief increase of NK cells in patients with infections was followed by decrease in all circulating lymphocytes. This is also true when persistent stress is induced on to the body. This study also demonstrated how the sympathetic nervous and immune system work together in the body, which sometimes is overlooked when one, is just looking at one specific item. It showed how hormones like norepinephrine and epinephrine are released during stresses, which decrease NK cells during cell adhesion to the vascular which then increase NK cells in peripheral blood. Even though this study is small as far is the number of people participating in the study, it does indicate there is a need for further study regarding the lymphocytes as biomarkers for infections.
The discovery of new processes by well-studied immune cells is remarkable. I decided to look into conditions required for macrophage-mediated repair and found this paper. In this article , they gather that monocytes and macrophages can change surface receptors depending on the levels of inflammation. Environments with no inflammation promoted macrophages which can undergo tissue repair. I thought this was fascinating and wondered what kind of therapeutic treatments this could lead to in the future.Unfortunately, I could not find much on possible treatment research. I did find an article that uses these macrophages in the treatment of inflammation and fibrosis. To summarize, researchers are looking to inject these altered cells into wound sites for increased wound healing. I imagine these cells would be used for in many inflammatory diseases and artery repair in heart attacks. I always get excited when new discoveries are made and look forward into the effect they have on the scientific world.
-Dakingindanorf
Who knew that macrophages of all cells could help repair blood vessels? This got me thinking about the major vessels in our bodies – the heart vessels. I came across a paper titled Macrophage-Stem Cell Crosstalk after Myocardial Infarction which talked about how macrophages mediate vessel repair in the heart. Essentially they induced heart attacks in mice and then injected either bone marrow mesenchymal stromal cells (MSCs), mononuclear cells, or saline into the infarct. All three of these solutions were also tested with and without macrophages. Their results showed that the MSC solution with macrophages markedly increased the amount of cytokine production necessary for vessel healing, compared to the results of the mononuclear cells and saline injection. This article was really eye opening because research towards macrophage and stem cell communication could be a breakthrough in speeding up the healing of a heart attack.
http://www.sciencedirect.com/science/article/pii/S0735109713031008
This post is very interesting. It is also important to the future of health because of the high prevalence of disease processes such as dementia, Alzheimer’s disease, and cerebrovascular accidents (stroke). Ruptured blood vessels cause increased bleeding in the brain, which can leading to blood clotting leading to ischemia, or cell death. Macrophages have been found to have a secondary function, of repairing ruptured blood vessels. The limiting factor here, however, is that only a single macrophage arrives to the site of the ruptured blood vessel for the repair, and takes 3 hours for complete repair of the blood vessel. It is random as well, because if 2 macrophages arrive at the site of the ruptured vessels, they will leave without repairing it.
What seems to be essential here is what is causing the recruit of the macrophage? When a blood vessel ruptures, there must be some signal or cytokine released to attract the macrophage to the site of the hemorrhage. The signal is probably very specific, as it has been seen that only one macrophage is recruited. Maybe potentially strengthening this signal, or increasing the amount of cytokine released, will recruit more macrophages. More macrophages being recruited to the area of a blood vessel will repair the vessel more efficiently, and in a timely manner. Time is essential when it comes to processes such as a stroke. Stopping a bleed early can decrease the severity of the disease.
Macrophages are known for their ability to surround and eat dead cells and anything else foreign and not protein specific in the body, through phagocytosis. Macrophages are among the first line of defense of pathogens invading human tissues; these cells can participate in both innate and adaptive immunity. However, the role of macrophages extends beyond their host defense against bacteria and parasites. So, it was not to my surprise that macrophages could play a role in mending broken blood vessels because further research they have found these cells to have an increasing number of activation states. Macrophages are known to patrol within the cavity, and once they sense damage of organs, tissues or blood vessels, they will act accordingly to initiate an immediate and rapid response. Macrophages are always present, so their quick response to repair the broken blood vessel was like a natural reflex. However, in the case of mending the blood vessels the macrophages are not seen to improve as quickly as they do at the site of an infection. The researchers observed that it took about three hours for the macrophage mending process to be complete and if macrophages are used for therapeutic use in dementia and cognitive deterioration it is important that they evaluate ways to speed up the repair process. There is so much to discover with these cells, but the evolving research is remarkable, and I hope one day this research with broken vessels enhances and used for therapeutic therapy for individuals who suffer from microbleeds that lead to severe medical complications.
In conclusion, I am curious to do further research on why only one macrophage is capable of mending the broken blood vessel. In my thought process if I would think that if more than one macrophages could work on the broken blood vessel, then the process would be faster but I could be wrong. I’ll be awaiting further research on that topic, personally.
Parham, P. (2014). The immune system. Garland Science.
I think it would be worthwhile to look into what certain cellular signaling pathway or soluble molecules (cytokines and chemokines) may cause the migration of these macrophages to the broken blood vessel site. It could be interesting to develop a human microglia culture in vitro much like the In vitro microvessels model. In theory, one could break the blood vessel, and analyze the macrophage components that have migrated. There is only one thing missing…the immune system. Perhaps a better way to understand what cytokines may cause the recruitment of these ligase-like macrophages could be to harvest and proliferate the macrophages in cell culture, apply different cytokines, and measure migratory action and/or proliferation of the cells. If this is successful, one could perhaps treat the adult with the cytokine to stimulate the macrophage function to repair the blood vessels and thus, prevent future neurological deterioration that accompanies aging. This however, could also prove complicated since many cytokines have multiple functions and an increase in any of the pool could have effects on other pathways. One may also consider macrophage cell based therapy, in which macrophages would be delivered to the patient. Macrophages are making headway in regenerative medicine and I think this could be truly promising for those suffering with brain microbleeds leading to dementia and related age disorders.
This is a very interesting article because when most people hear or think of macrophage, it is commonly known as a phagocyte in the innate immune response. But due to this article it seems that even with the human body as we live and breathe there are still many things that we are unaware of. I feel that this information could be harvested along with further research in the to deal with problems that humans are plagued with such as myocardial infarct due to a ischemic stroke. In my research of this topic I came over this article entitled “Cannabinoid Type 2 Receptor Activation Downregulates Stroke-Induced Classic and Alternative Brain Macrophage/Microglial Activation Concomitant to Neuroprotection”, it is an experiment in which scientists examined the effects of CB2R (Cannabinoid Type 2 Receptor) agonist on infarct size. The experimentation showed that introduction of the agonist JWH-133 decreased the infarct size of a middle cerebral artery occlusion. I feel this topic could lead to an age in where what is now thought impossible will be possible, such as reconnecting severed limbs or vascularizing prosthesis.
Link to article